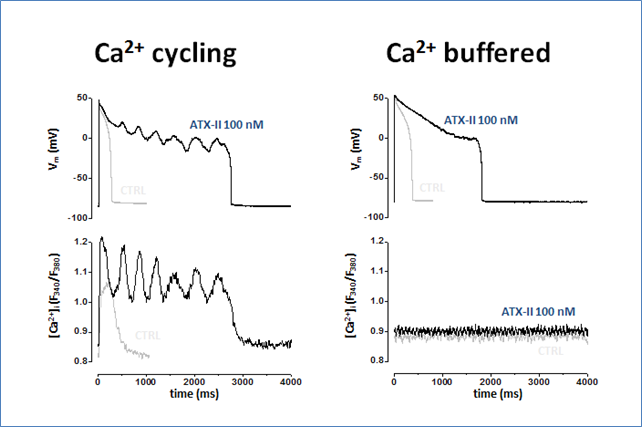
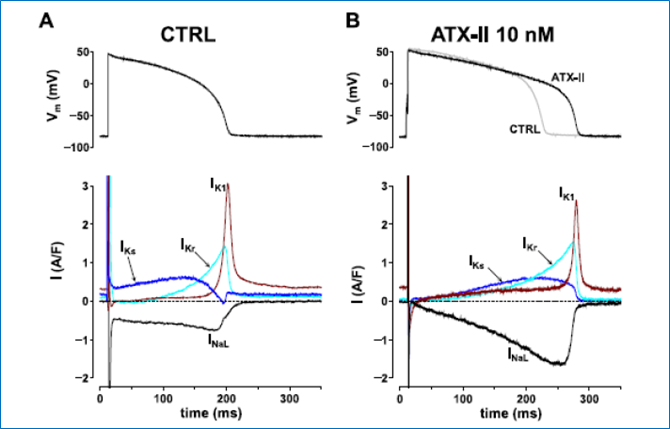
To study the feedback interaction between Ca2+ and ionic currents, we further developed the selfAP-clamp with Ca2+ cycling technique, which preserves Ca2+ homeostasis and beat-to-beat contraction under AP-clamp by matching the internal and external physiological solutions. This approach enables studying how Ca2+ transient modifies various Ca2+-sensitive channels to reshape the action potential and what happens under disease conditions. We discovered that the late Na+ current (INaL) recorded under selfAP-clamp is significantly larger than previously predicted from V-clamp data; this Na+ influx during AP plateau phase can substantially elevate the intracellular [Na+] to cause Ca2+ overload, which results in spontaneous Ca2+ oscillations leading to EADs and DADs. These findings reveal the dual roles of INa in altering APD as an inward current and also altering Na+-Ca2+ homeostasis as a substantial Na+ influx.
 |
 |
Relevant Publications
- Tamas Banyasz, Balazs Horvath, Zhong Jian, Leighton T. Izu, Chen-Izu Y*. Profile of L-type Ca2+ current and Na+/Ca2+ exchange current during a cardiac action potential in ventricular myocytes. Heart Rhythm (2012) 9:134-142. PMID: 21884673 PMC3252888
- Horvath B, Banyasz T, Jian Z, Hegyi B, Kistamas K, Nanasi PP, Izu LT, Chen-Izu Y*. Dynamics of the late Na+ current during cardiac action potential and its contribution to afterdepolarizations. J Mol Cell Cardiol. (2013) 64C:59-68. PMID: 24012358 PMC3856763
